Why Label Literacy Matters
Sunscreen is a cornerstone of skin‑health strategy because it blocks ultraviolet radiation that causes sunburn, premature aging and skin cancer. Understanding the label helps patients choose products that truly protect. Frequently seen terms can be misleading: “waterproof” is prohibited by the FDA, which instead uses “water‑resistant” (40 min) or “very water‑resistant” (80 min); “sports” and “baby” have no regulatory definition and may vary in ingredient composition. The most critical claim is “broad‑spectrum.” This designation, required by FDA testing, confirms that the product shields against both UVB (burning) and UVA (aging) rays, reducing DNA damage and long‑term cancer risk. Selecting a broad‑spectrum sunscreen with SPF 30 or higher and applying it liberally maximizes protection and supports overall skin wellness. Patients should reapply two hours, especially after swimming, sweating, or towel‑drying, to maintain label’s promised protection.
What “Broad‑Spectrum” Actually Means
| Aspect | Details |
|---|---|
| FDA Definition | Passed critical‑wavelength test (≥ 370 nm) → protects both UVA and UVB |
| UVA (aging) | ~95 % of UV radiation; causes premature aging, DNA damage, skin cancer |
| UVB (burning) | Causes sunburn; also contributes to skin cancer risk |
| Label cue | Words “Broad Spectrum” on front panel, usually with SPF ≥ 30 |
| PA rating | Used outside U.S.; grades UVA protection (PA+, PA++, PA+++). Broad‑spectrum confirms minimum UVA coverage but does not quantify it |
 In the United States the FDA defines a “broad‑spectrum” sunscreen as a product that has passed a critical‑wavelength test (≥ 370 nm) and therefore protects against both UVA (aging, deeper‑penetrating) and UVB (burning) rays. UVA accounts for roughly 95 % of UV radiation reaching the earth’s surface, drives premature aging, DNA damage and contributes to skin cancer, while UVB causes sunburn and also plays a role in cancer risk.
In the United States the FDA defines a “broad‑spectrum” sunscreen as a product that has passed a critical‑wavelength test (≥ 370 nm) and therefore protects against both UVA (aging, deeper‑penetrating) and UVB (burning) rays. UVA accounts for roughly 95 % of UV radiation reaching the earth’s surface, drives premature aging, DNA damage and contributes to skin cancer, while UVB causes sunburn and also plays a role in cancer risk.
Difference between broad‑spectrum and regular sunscreen – Regular sunscreen, lacking the broad‑spectrum claim, is formulated mainly to block UVB radiation. It may prevent sunburn but leaves skin vulnerable to UVA‑induced photo‑damage. Broad‑spectrum sunscreen offers more complete daily protection by covering both spectra.
Identifying the claim on a label – Look for the words “Broad Spectrum” printed on the front panel, often paired with an SPF number (≥ 30). The claim signals FDA‑approved UVA protection. Some products also display a PA rating (PA+, PA++, PA+++ or PA++) or a UVA‑circle logo, which provides further detail on the strength of UVA defense.
Broad‑spectrum vs. PA rating – The FDA’s broad‑spectrum label confirms adequate UVA coverage, but it does not quantify it. The PA rating, used mainly outside the U.S., grades UVA protection with plus signs; a high PA rating (PA+++ or PA++) indicates stronger UVA defense than the minimum required for broad‑spectrum status.
Mineral, Chemical, and Hybrid Formulas
| Formula type | Typical actives | Key traits | Ideal skin type |
|---|---|---|---|
| Mineral (physical) | Zinc oxide, Titanium dioxide | Reflects UV, fragrance‑free, good for sensitive/baby skin | Sensitive, infants |
| Chemical (organic) | Avobenzone, Octocrylene, Oxybenzone, etc. | Absorbs UV, lighter feel, can be non‑comedogenic with dimethicone/silica | Oily, acne‑prone (when non‑comedogenic) |
| Hybrid | Combination of mineral + chemical actives | Stability of zinc oxide + cosmetic elegance of organics | Users wanting best of both worlds |
| Oily‑skin recommendation | Broad‑spectrum, non‑comedogenic, matte‑finishing agents (silica, dimethicone) + SPF ≥ 30 | Controls shine, avoids heavy emollients | |
| Water‑resistance mechanism | UV filters + film‑forming polymers (e.g., VP/eicosene copolymer, dimethicone) + stabilizers (aloe, glycerin) | SPF holds for 40 or 80 min of swimming/sweating |
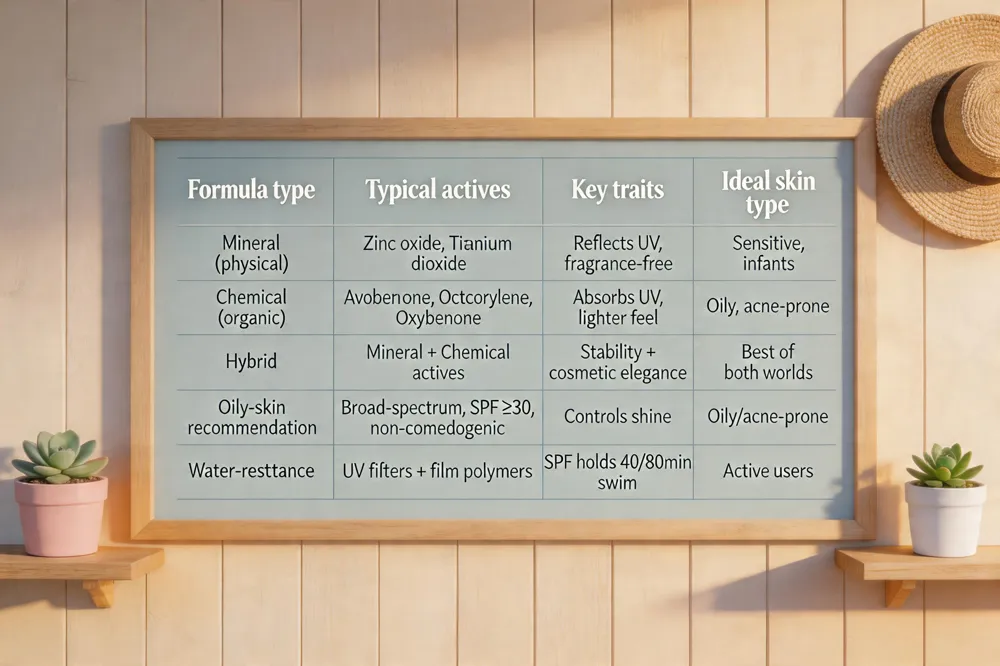 Broad‑spectrum protection is a labeling claim, not an ingredient class. Both mineral (physical) sunscreens—using zinc oxide, titanium dioxide, or both—and chemical (organic) sunscreens—using filters such as avobenzone, octocrylene, or oxybenzone—can meet the FDA’s critical‑wavelength test and be labeled broad‑spectrum.
Broad‑spectrum protection is a labeling claim, not an ingredient class. Both mineral (physical) sunscreens—using zinc oxide, titanium dioxide, or both—and chemical (organic) sunscreens—using filters such as avobenzone, octocrylene, or oxybenzone—can meet the FDA’s critical‑wavelength test and be labeled broad‑spectrum.
Physical sunscreens form a reflective barrier and are generally recommended for sensitive or baby skin because they lack fragrance, oils, and many allergens. Chemical sunscreens absorb UV photons and are often lighter‑feel, but some people with acne‑prone or oily skin prefer non‑comedogenic, oil‑free formulas that contain dimethicone, silica, or polymeric film formers to control shine. Hybrid products combine mineral and chemical actives, leveraging the stability of zinc oxide with the cosmetic elegance of organic filters.
For oily skin, look for broad‑spectrum, non‑comedogenic options that include matte‑finishing agents (e.g., silica, dimethicone) and a high SPF (≥30) without heavy emollients.
Water‑resistant sunscreens achieve durability by pairing UV filters with film‑forming polymers (VP/eicosene copolymer, dimethicone) and stabilizers such as aloe vera or glycerin, allowing the SPF to hold for 40 or 80 minutes of swimming or sweating.
Importantly, no sunscreen is truly waterproof; the FDA prohibits the term “waterproof.” Products are labeled “water‑resistant” for a specific duration, and reapplication is required after that time, after towel‑drying, or heavy sweating.
U.S. Sunscreen Label Regulations (2020‑2022)
| Requirement | Details |
|---|---|
| Regulatory basis | FDA OTC monograph M020 (21 CFR Part 352) |
| Active ingredients | Listed alphabetically with percentages; must be permitted filters |
| SPF minimum | ≥ 2; “Broad Spectrum” claim requires critical‑wavelength ≥ 370 nm |
| Principal display panel | Must show “Sunscreen”, dosage form, SPF number, optional “Broad Spectrum”, water‑resistance claim (40 min or 80 min) |
| Prohibited terms | “Waterproof”, “Sweatproof”, “Sunblock” |
| Safety warnings | External use only; do not apply on broken skin; children < 6 months – not for use |
| Water‑resistance labeling | “Water‑Resistant” (40 min) or “Very Water‑Resistant” (80 min) – must reapply after swimming, sweating, or towel‑drying |
 FDA OTC monograph and 21 CFR Part 352
Sunscreens are classified as over‑the‑counter drugs under the FDA’s OTC monograph M020 (21 CFR Part 352). The monograph lists permitted active ingredients—zinc oxide, titanium dioxide, avobenzone, octocrylene, oxybenzone, etc.—and sets concentration limits. Each product must achieve a minimum SPF of 2 and, if it wishes a “Broad Spectrum” claim, must pass the critical‑wavelength test (≥ 370 nm).
FDA OTC monograph and 21 CFR Part 352
Sunscreens are classified as over‑the‑counter drugs under the FDA’s OTC monograph M020 (21 CFR Part 352). The monograph lists permitted active ingredients—zinc oxide, titanium dioxide, avobenzone, octocrylene, oxybenzone, etc.—and sets concentration limits. Each product must achieve a minimum SPF of 2 and, if it wishes a “Broad Spectrum” claim, must pass the critical‑wavelength test (≥ 370 nm).
Label elements required on the principal display panel The principal display panel must show the identity “Sunscreen,” the dosage form, the SPF number, and the phrase “Broad Spectrum” only when the UVA test is met. Active ingredients and their percentages are listed alphabetically, and the water‑resistance claim ("Water‑Resistant" 40 min or "Very Water‑Resistant" 80 min) appears in the same line with uniform font.
Water‑resistance claims and prohibited terms The FDA prohibits the terms “waterproof,” “sweatproof,” and “sunblock.” Labels may state a 40‑ or 80‑minute water‑resistance claim and must advise reapplication after swimming, sweating, or towel‑drying.
Warnings for children and external use All labels include a warning that the product is for external use only, a “Do not use on damaged or broken skin” notice, and a specific statement that children under six months should not use sunscreen.
Key Q&A
- Sunscreen labeling regulations USA 2022: Products follow the 2011 final rule; they must list active ingredients, SPF, broad‑spectrum status, water‑resistance duration, usage directions, and child‑use warnings, all in English and in the FDA‑mandated format.
- Sunscreen labeling regulations USA 2021: The same 2011 rule applied; the principal display panel must begin with “Sunscreen,” show SPF, broad‑spectrum and water‑resistance claims, and include alphabetical ingredient listings and child‑use cautions.
- Sunscreen labeling regulations USA 2020: Under the monograph, labels required active ingredients, SPF, broad‑spectrum claim (if qualified), water‑resistance statement, use directions, external‑use warning, and a note that infants under six months should not apply the product.
- FDA sunscreen labeling regulations USA: 21 CFR Part 352 mandates the principal display panel to convey product name, SPF, broad‑spectrum status, water‑resistance claim, and active ingredients; the information panel adds directions, external‑use warning, and the six‑month‑old child restriction.
- Sunscreen monograph FDA: Monograph M020 defines permitted filters, concentration limits, the requirement that each ingredient contribute at least SPF 2, and that the overall SPF be at least twice the number of active ingredients; broad‑spectrum labeling requires the critical‑wavelength test.
- FDA sunscreen regulations 2025: The agency proposed adding bemotrizinol as a new broad‑spectrum active ingredient, reflecting the first amendment to the monograph in 25 years and signalling a move toward modernized testing standards.
SPF Selection, Application Amounts, and Re‑application
| Topic | Guidance |
|---|---|
| What SPF measures | UVB protection only; SPF 30 ≈ 97 % UVB blocked, SPF 50 ≈ 98 %, SPF 70 ≈ 98.5 % |
| Diminishing returns | Little added benefit beyond SPF 30; focus on proper amount & re‑application |
| Application amount | Full body: ~1 oz (shot‑glass); Face & neck: ~¼ tsp (≈ 2 finger‑lengths) |
| Re‑application interval | Every 2 hours; sooner after swimming, sweating, or towel‑drying |
| Adapalene users | Broad‑spectrum SPF 30+ applied 15–30 min before exposure; SPF 50+ for very fair/photosensitive skin |
| Choosing SPF for face | Minimum SPF 30 for daily use; SPF 50 for prolonged outdoor time or fair skin |
 Broad‑spectrum sunscreen is the cornerstone of daily skin protection because it blocks both UVA (aging) and UVB (burn) rays. SPF measures only UVB protection; SPF 30 blocks ~97 % of UVB, SPF 50 ~98 %, and SPF 70 ~98.5 %. The incremental benefit diminishes sharply after SPF 30, so proper application and re‑application matter more than a higher number.
Broad‑spectrum sunscreen is the cornerstone of daily skin protection because it blocks both UVA (aging) and UVB (burn) rays. SPF measures only UVB protection; SPF 30 blocks ~97 % of UVB, SPF 50 ~98 %, and SPF 70 ~98.5 %. The incremental benefit diminishes sharply after SPF 30, so proper application and re‑application matter more than a higher number.
What SPF numbers really mean – SPF is the factor by which sunburn time is multiplied compared with unprotected skin.
Diminishing returns above SPF 30 – Going from SPF 30 to 50 adds only about 1 % more UVB blockage; SPF 70 adds another ~0.5 %.
How much product to use – Apply about one ounce (a shot‑glass) for full‑body coverage, or roughly a quarter‑teaspoon (≈ 2 finger‑lengths) for the face and neck.
Re‑application timing for optimal protection – Reapply every two hours, and sooner after swimming, sweating, or towel‑drying; water‑resistant labels indicate protection for 40 or 80 minutes of water exposure.
Is SPF 30 enough when using adapalene? Yes, a broad‑spectrum SPF 30+ sunscreen should be applied 15‑30 minutes before sun exposure and reapplied every two hours; SPF 50+ is advisable for very fair or highly photosensitive skin.
What is better sunscreen, 50 or 70? Both are broad‑spectrum; SPF 50 blocks ~98 % of UVB, SPF 70 ~98.5 %. The modest extra protection of SPF 70 is mainly useful for highly fair, photosensitive, or high‑risk patients.
Which SPF is best for the face? At least SPF 30 is recommended for everyday use; SPF 50 offers a safety margin for prolonged outdoor time or very fair skin.
How to read a sunscreen label? Look for the “broad‑spectrum” claim, SPF ≥ 30, water‑resistance (40 or 80 minutes), active ingredient type (chemical vs. mineral), and clear usage directions.
Family‑Friendly Water‑Resistant Options
| Product type | Example (SPF) | Water‑resistance | Key features |
|---|---|---|---|
| Adult face | EltaMD UV Pure Broad‑Spectrum SPF 47 | 80 min | Lightweight, non‑comedogenic |
| Adult face (fragrance‑free)** | Supergoop! PLAY Everyday Lotion SPF 50 | 80 min | |
| Mineral‑only | Badger Adventure Mineral Sunscreen Cream SPF 50 | 40 min | Reef‑friendly, zinc oxide/titanium dioxide |
| Kids (mineral) | Blue Lizard Kids Mineral SPF 50 Spray | 80 min | Fragrance‑free, gentle on sensitive skin |
| Kids (hypoallergenic) | SolRX Kids Sunscreen | 80 min | Tear‑free, reef‑safe |
| Kids (clear spray) | Sun Bum Kids SPF 50 Clear Sunscreen Spray | 80 min | Lightweight finish |
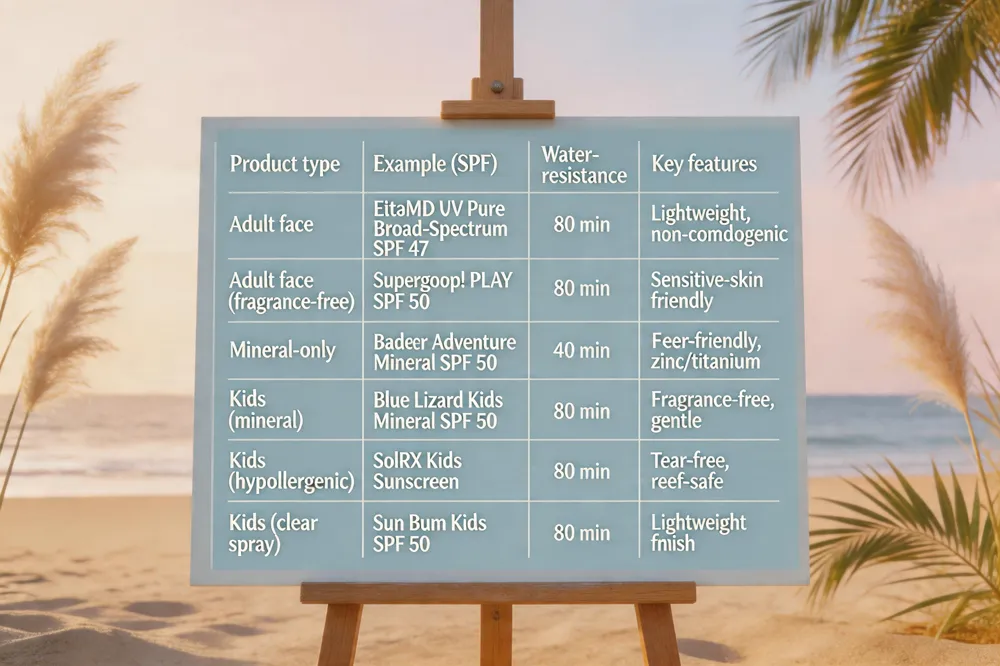 The FDA permits only the terms “water‑resistant” (40 minutes) or “very water‑resistant” (80 minutes) on sunscreen labels; products must retain their labeled SPF after the specified immersion time and be reapplied after swimming, sweating, or towel‑drying. For children, choose a “broad‑spectrum sunscreen” with “SPF 30 or higher”, an “80‑minute resistance rating”, and gentle, “fragrance‑free” mineral filters (“zinc oxide or titanium dioxide”) that are less likely to irritate delicate skin.
The FDA permits only the terms “water‑resistant” (40 minutes) or “very water‑resistant” (80 minutes) on sunscreen labels; products must retain their labeled SPF after the specified immersion time and be reapplied after swimming, sweating, or towel‑drying. For children, choose a “broad‑spectrum sunscreen” with “SPF 30 or higher”, an “80‑minute resistance rating”, and gentle, “fragrance‑free” mineral filters (“zinc oxide or titanium dioxide”) that are less likely to irritate delicate skin.
Best water‑resistant sunscreen for face – Dermatologists often recommend EltaMD UV Pure Broad‑Spectrum SPF 47, a lightweight, non‑comedogenic formula that stays effective for up to “80 minutes” in water. Supergoop!’s PLAY Everyday Lotion SPF 50 offers “fragrance‑free”, “80‑minute water resistance” for sensitive skin, while Badger Adventure Mineral Sunscreen Cream SPF 50 provides “reef‑friendly” protection with a “40‑minute rating” for mineral‑only lovers.
Water‑resistant sunscreen for kids – Look for “broad‑spectrum” SPF 30 + and “80‑minute resistance”. Blue Lizard Kids Mineral SPF 50 Spray uses “zinc oxide”, is “fragrance‑free”, and gentle on sensitive skin. SolRX Kids Sunscreen offers an 8‑hour, tear‑free, “hypoallergenic”, “reef‑safe” formula, and Sun Bum’s Kids SPF 50 Clear Sunscreen Spray delivers “80‑minute water resistance” with a lightweight finish.
Ingredient safety for sensitive skin – “Physical (mineral) sunscreens” containing “zinc oxide or titanium dioxide” are generally recommended for sensitive or infant skin because they reflect UV rays and lack irritating fragrances, oils, or PABA. “Chemical filters” can cause allergies in some users, so a “fragrance‑free”, “hypoallergenic” label is a key safety cue.
Beyond the Bottle: Complementary Sun‑Safety Practices
| Practice | Why it matters |
|---|---|
| Shade & UPF clothing | UPF 50 fabrics block ~98 % UV; reduces sunscreen needed and provides continuous protection |
| Separate repellent | Apply sunscreen first, wait 15 min, then insect repellent to avoid dilution/irritation |
| Infant protection | For < 6 months: shade, clothing, avoid direct sun; after 6 months use mineral sunscreen only |
| Reef‑safe choices | Avoid oxybenzone & octinoxate; mineral sunscreens (zinc oxide/titanium dioxide) are less harmful to coral |
| Label reading order | 1️⃣ Broad‑Spectrum claim → 2️⃣ SPF ≥ 30 → 3️⃣ Water‑resistance → 4️⃣ Active ingredient type |
 Protecting skin isn’t limited to the tube of sunscreen you apply.
Protecting skin isn’t limited to the tube of sunscreen you apply.
Shade, clothing, and UPF garments – The American Academy of Dermatology stresses that broad‑spectrum sunscreen with SPF 30+ should be paired with physical barriers. Wide‑brimmed hats, tightly‑woven shirts, and pants made from fabrics rated UPF 50 block about 98 % of UV radiation, reducing the amount of sunscreen needed and providing continuous protection even when re‑application is missed.
Avoid sunscreen‑insect‑repellent combos – The AAD advises using separate products; combining them can dilute each ingredient’s efficacy and increase the risk of skin irritation. Apply sunscreen first, wait 15 minutes, then layer repellent for optimal coverage against both UV rays and insects.
Infants and babies – For children under 6 months, shade, protective clothing, and avoidance of direct sun are preferred. Once a child is older than 6 months, choose a mineral sunscreen containing only zinc oxide or titanium dioxide, which is less likely to irritate delicate skin.
Environmental concerns and reef‑safe choices – “Reef‑safe” labels are unregulated, but mineral sunscreens that omit oxybenzone and octinoxate are generally considered less harmful to coral ecosystems. Selecting such products supports both personal skin health and marine preservation.
How to read a sunscreen label? – Look first for the “broad‑spectrum” claim (UVA + UVB protection), then an SPF of 30 or higher. Confirm water‑resistance (40 min / 80 min) and note the active ingredient type—mineral (zinc oxide, titanium dioxide) or chemical (avobenzone, octocrylene).
Sunscreen labeling regulations USA 2020 – Sunscreens are regulated as OTC drugs under the FDA monograph. Labels must list active ingredients, concentrations, SPF, a broad‑spectrum statement if the product passes the critical‑wavelength test, and any water‑resistance claim. The 2020 CARES Act introduced an administrative‑order pathway for updating these rules, but the core 2011 requirements remain in effect.
Putting It All Together
Understanding sunscreen labels empowers you to choose the protection your skin truly needs. Look for a broad‑spectrum claim, an SPF of 30 or higher, and a water‑resistance designation of 40 or 80 minutes if you’ll be active or in water. Note whether the product is mineral (zinc oxide/titanium dioxide), chemical, or hybrid, and check for “sensitive‑skin” or “baby” wording if irritation is a concern. The most important habit, however, is consistent daily application—about one ounce for the whole body or a teaspoon for the face—applied 15 minutes before exposure and reapplied every two hours, or sooner after swimming, sweating, or towel‑drying. If you have a history of skin cancer, unusual reactions to sunscreen, or need guidance for infants, children, or specific skin conditions, schedule a dermatology visit for recommendations.
